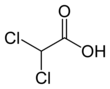
Dichloroacetic acid
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Dichloroacetic acid | |||
| Other names
Dichloroethanoic acid, bichloroacetic acid, DCA, BCA, dichloracetic acid, bichloracetic acid
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 1098596 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.001.098 | ||
| EC Number |
| ||
| 2477 | |||
| KEGG | |||
| MeSH | Dichloroacetate | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1764 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H2Cl2O2 | |||
| Molar mass | 128.94 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Density | 1.5634 g/cm3 (20 °C) | ||
| Melting point | 9 to 11 °C (48 to 52 °F; 282 to 284 K) | ||
| Boiling point | 194 °C (381 °F; 467 K) | ||
| miscible | |||
| Solubility | miscible with ethanol, diethyl ether[1] | ||
| Acidity (pKa) | 1.35[1] | ||
| -58.2·10−6 cm3/mol | |||
| Thermochemistry | |||
Std enthalpy of
formation (ΔfH⦵298) |
−496.3 kJ·mol−1[1] | ||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Warning | |||
| H314, H400 | |||
| P260, P264, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P391, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | MSDS (jtbaker) | ||
| Related compounds | |||
Related chloroacetic acids
|
Chloroacetic acid Trichloroacetic acid | ||
Related compounds
|
Acetic acid Difluoroacetic acid Dibromoacetic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Dichloroacetic acid (DCA), sometimes called bichloroacetic acid (BCA), is the organic compound with formula CHCl2CO2H. It is an analogue of acetic acid, in which 2 of the 3 hydrogen atoms of the methyl group have been replaced by chlorine atoms. Like the other chloroacetic acids, it has various practical applications. The salts and esters of dichloroacetic acid are called dichloroacetates.
Reactions
[edit]The chemistry of dichloroacetic acid is typical for halogenated organic acids. It is an alkylating agent. It forms esters.
It is a member of the chloroacetic acids family. As such it is more acidic than acetic acid. It fully dissociates into dichloroacetate when dissolved in water, consistent with it pKa of 1.35,[1] pure dichloroacetic acid is classed as a strong organic acid; it is very corrosive and extremely destructive to tissues of the mucous membranes and upper respiratory tract via inhalation.[2]
Natural occurrence
[edit]DCA has been shown to occur in nature in at least one seaweed, Asparagopsis taxiformis [3] and also in the mushroom Russula nigricans.[4] It is a trace product of the chlorination of drinking water and is produced by the metabolism of various chlorine-containing drugs or chemicals.[5] DCA is typically prepared by the reduction of trichloroacetic acid (TCA).[6] DCA is prepared from chloral hydrate also by the reaction with calcium carbonate and sodium cyanide in water followed by acidifying with hydrochloric acid.[7]
As a laboratory reagent, both DCA and TCA[8] are used as precipitants to prompt macromolecules such as proteins to precipitate out of solution.[9]
Therapeutic uses
[edit]Salts of DCA have been studied as potential drugs because they inhibit the enzyme pyruvate dehydrogenase kinase.[10] Although preliminary studies found that DCA can slow the growth of certain tumors in animal studies and in vitro studies, as of 2012 insufficient evidence supported the use of DCA for cancer treatment.[11]
Lactic acidosis
[edit]A randomized controlled trial in children with congenital lactic acidosis found that while DCA was well tolerated, it was ineffective in improving clinical outcomes.[12] A separate trial of DCA in children with MELAS (a syndrome of inadequate mitochondrial function, leading to lactic acidosis) was halted early, as all 15 of the children receiving DCA experienced significant nerve toxicity without any evidence of benefit from the medication.[13] A randomized controlled trial of DCA in adults with lactic acidosis found that while DCA lowered blood lactate levels, it had no clinical benefit and did not improve hemodynamics or survival.[14]
Thus, while early case reports and pre-clinical data suggested that DCA might be effective for lactic acidosis, subsequent controlled trials have found no clinical benefit of DCA in this setting. In addition, clinical trial subjects were incapable of continuing on DCA as a study medication owing to progressive toxicities.
Cancer
[edit]In 2007 reports emerged in the press and via the Internet that Evangelos Michelakis and coworkers at the University of Alberta had reported that sodium dichloroacetate reduced tumors in rats and killed cancer cells in vitro.[15] Because the drug cannot be patented, financing the broad and expensive testing required to obtain FDA approval is problematic.[16] The US Food and Drug Administration enforces the law that prohibits the sale of substances with the suggestion that they are cancer treatments unless they have been approved by the FDA.[17]
The American Cancer Society in 2012 stated that "available evidence does not support the use of DCA for cancer treatment at this time."[11] Physicians warned of potential problems if people attempt to try DCA outside a controlled clinical trial.[18] One problem with attempting this is obtaining the chemical. One fraudster was sentenced to 33 months in prison for selling a white powder containing starch, but no DCA, to people with cancer.[19]
The only monitored in vivo dosage of five human patients with glioblastoma with DCA was not designed to test its efficacy against their cancer. This study was rather to see whether it could be given at a specific dosage safely without causing side effects (e.g. neuropathy). All five patients were receiving other treatments during the study.[20][21] Observations in vitro and of tumours extracted from those five patients suggest that DCA might act against cancer cells by depolarising abnormal mitochondria found in glioblastoma cancer cells – allowing the mitochondria to induce apoptosis (cell death) of the malignant cells.[20] In vitro work with DCA on neuroblastomas (which have fewer recognised mitochondrial abnormalities) showed activity against malignant, undifferentiated cells.[22] A 2016 case report discussed and reviewed the application of DCA in central nervous system malignancies.[23] A 2018 study found that DCA could trigger a metabolic switch from glycolysis (the Warburg effect) to mitochondrial OXPHOS and increase reactive oxygen stress affecting tumor cells. These effects were not observed in non-tumor cells.[24]
Neuropathy
[edit]Neuropathy has been a problem in some clinical trials with DCA causing them to be effectively halted,[13] but a 2008 BJC review found that it has not occurred in other DCA trials.[25] The mechanism of DCA induced neuropathy is not well understood.[26] On the one hand in vitro work with nerves has suggested a mechanism for the neuropathic effect of DCA; with DCA showing a dose and exposure dependent demyelination of nerves (stripping of the nerve 'sheath'), which demyelination was partially reversible over time, following washout of DCA.[27] On the other hand, the 2008 review in BJC [25] states "This neurotoxicity resembled the pattern of length-dependent, axonal, sensorimotor polyneuropathy without demyelination." with regard to the 2006 study by Kaufman et al.[13]
Heart failure
[edit]DCA has been investigated as a treatment for post-ischemic recovery.[28] There is also evidence that DCA improves metabolism by NADH production stimulation, but may lead to a depletion of NADH in normoxia.[29]
See also
[edit]- Dalapon (dichloropropionic acid)
References
[edit]- ^ a b c d Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. ISBN 1-4398-5511-0.
- ^ "Dichloroacetic Acid". Hazard.com. 1998-04-21. Retrieved 2015-04-17.
- ^ [1] Archived April 16, 2015, at the Wayback Machine
- ^ Lajin, B.; Braeuer, S.; Borovička, J.; Goessler, W. (2021). "Is the water disinfection by-product dichloroacetic acid biosynthesized in the edible mushroom Russula nigricans?". Chemosphere. 281: 130819. Bibcode:2021Chmsp.28130819L. doi:10.1016/j.chemosphere.2021.130819. PMID 33991903.
- ^ Stacpoole, Peter W.; Henderson, George N.; Yan, Zimeng; James, Margaret O. (1998). "Clinical Pharmacology and Toxicology of Dichloroacetate". Environmental Health Perspectives. 106 (Suppl 4): 989–94. doi:10.1093/jnci/djx071. JSTOR 3434142. PMC 1533324. PMID 9703483.
- ^ Doughty, Howard Waters.; Derge, Gerhard Julius. (1931). "Preparation of Dichloroacetic Acid". Journal of the American Chemical Society. 53 (4): 1594–1596. doi:10.1021/ja01355a066. ISSN 0002-7863.
- ^ "Dichloroacetic Acid". Organic Syntheses. 19: 38. 1939. doi:10.15227/orgsyn.019.0038.
- ^ Koontz, Laura (2014). "TCA precipitation". Laboratory Methods in Enzymology: Protein Part C. Vol. 541. pp. 3–10. doi:10.1016/B978-0-12-420119-4.00001-X. ISBN 978-0-12-420119-4. ISSN 1557-7988. PMID 24674058.
- ^ Rajalingam, Dakshinamurthy; Loftis, Charles; Xu, Jiashou J; Kumar, Thallapuranam Krishnaswamy S (2009). "Trichloroacetic acid-induced protein precipitation involves the reversible association of a stable partially structured intermediate". Protein Science. 18 (5): 980–993. doi:10.1002/pro.108. ISSN 0961-8368. PMC 2771300. PMID 19388015.
- ^ Stacpoole, Peter W. (2017). "Therapeutic Targeting of the Pyruvate Dehydrogenase Complex/Pyruvate Dehydrogenase Kinase (PDC/PDK) Axis in Cancer". JNCI: Journal of the National Cancer Institute. 109 (11). doi:10.1093/jnci/djx071. PMID 29059435.
- ^ a b "Dichloracetate (DCA)". American Cancer Society. Archived from the original on 3 May 2015. Retrieved 1 December 2012.
- ^ Stacpoole, P. W.; Kerr, D. S.; Barnes, C; Bunch, S. T.; Carney, P. R.; Fennell, E. M.; Felitsyn, N. M.; Gilmore, R. L.; Greer, M; Henderson, G. N.; Hutson, A. D.; Neiberger, R. E.; O'Brien, R. G.; Perkins, L. A.; Quisling, R. G.; Shroads, A. L.; Shuster, J. J.; Silverstein, J. H.; Theriaque, D. W.; Valenstein, E (2006). "Controlled Clinical Trial of Dichloroacetate for Treatment of Congenital Lactic Acidosis in Children". Pediatrics. 117 (5): 1519–31. doi:10.1542/peds.2005-1226. PMID 16651305. S2CID 38328451.
- ^ a b c Kaufmann, P.; Engelstad, K.; Wei, Y.; Jhung, S.; Sano, M. C.; Shungu, D. C.; Millar, W. S.; Hong, X.; Gooch, C. L.; Mao, X.; Pascual, J. M.; Hirano, M.; Stacpoole, P. W.; Dimauro, S.; De Vivo, D. C. (2006). "Dichloroacetate causes toxic neuropathy in MELAS: A randomized, controlled clinical trial". Neurology. 66 (3): 324–30. doi:10.1212/01.wnl.0000196641.05913.27. PMID 16476929. S2CID 19623200.
- ^ Stacpoole, Peter W.; Wright, Elizabeth C.; Baumgartner, Thomas G.; Bersin, Robert M.; Buchalter, Scott; Curry, Stephen H.; Duncan, Charles A.; Harman, Eloise M.; Henderson, George N.; Jenkinson, Steven; Lachin, John M.; Lorenz, Anthea; Schneider, Stephen H.; Siegel, John H.; Summer, Warren R.; Thompson, Douglas; Wolfe, Christopher L.; Zorovich, Barbara (1992). "A Controlled Clinical Trial of Dichloroacetate for Treatment of Lactic Acidosis in Adults". New England Journal of Medicine. 327 (22): 1564–9. doi:10.1056/NEJM199211263272204. PMID 1435883.
- ^ Andy Coghlan (Jan 17, 2007). "Cheap, 'safe' drug kills most cancers". New Scientist. Archived from the original on June 12, 2019. The original article did not have quotation marks around the word "safe".
- ^ "No patent? No cancer drug development". New Scientist. Jan 17, 2007.
- ^ "'Cancer drug' site shut down". New Scientist. Jul 25, 2007.
- ^ Andrea Sands (March 18, 2007). "Patients tout database for drug treatment". Edmonton Journal. p. A13.
"If it starts going badly, who is following you before it gets out of control? By the time you realize your liver is failing, you're in big trouble", said Laura Shanner, Associate Professor of Health Ethics at the University of Alberta.
- ^ "Canadian Man Sentenced to 33 Months in Prison for Selling Counterfeit Cancer Drugs Using the Internet". US Department of Justice. 25 August 2010. Retrieved January 5, 2018.
- ^ a b Michelakis, E. D.; Sutendra, G.; Dromparis, P.; Webster, L.; Haromy, A.; Niven, E.; Maguire, C.; Gammer, T. L.; MacKey, J. R.; Fulton, D.; Abdulkarim, B.; McMurtry, M. S.; Petruk, K. C. (2010). "Metabolic Modulation of Glioblastoma with Dichloroacetate". Science Translational Medicine. 2 (31): 31ra34. doi:10.1126/scitranslmed.3000677. PMID 20463368. S2CID 616711.
- ^ "Potential Cancer Drug DCA Tested in Early Trials". Archived from the original on February 18, 2011.
- ^ Vella, Serena; Conti, Matteo; Tasso, Roberta; Cancedda, Ranieri; Pagano, Aldo (2012). "Dichloroacetate inhibits neuroblastoma growth by specifically acting against malignant undifferentiated cells". International Journal of Cancer. 130 (7): 1484–93. doi:10.1002/ijc.26173. PMID 21557214. S2CID 19111145.
- ^ Lemmo, W; Tan, G (2016). "Prolonged Survival After Dichloroacetate Treatment of Non-Small-Cell Lung Carcinoma-Related Leptomeningeal Carcinomatosis". Journal of Medical Cases. 7 (4): 136–142. doi:10.14740/jmc2456w.
- ^ Zhou, Li; Liu, Lianlian; Chai, Wei; Zhao, Ting; Jin, Xin; Guo, Xinxin; Han, Liying; Yuan, Chunli (February 2019). "Dichloroacetic acid upregulates apoptosis of ovarian cancer cells by regulating mitochondrial function". OncoTargets and Therapy. 12: 1729–1739. doi:10.2147/ott.s194329. PMC 6419601. PMID 30881027.
- ^ a b Michelakis, E D; Webster, L; MacKey, J R (2008). "Dichloroacetate (DCA) as a potential metabolic-targeting therapy for cancer". British Journal of Cancer. 99 (7): 989–94. doi:10.1038/sj.bjc.6604554. PMC 2567082. PMID 18766181.
- ^ "Complementary Approaches: Dichloroacetate (DCA)". CancerQuest. Archived from the original on April 6, 2015.
- ^ Felitsyn, Natalia; Stacpoole, Peter W.; Notterpek, Lucia (2007). "Dichloroacetate causes reversible demyelination in vitro: Potential mechanism for its neuropathic effect". Journal of Neurochemistry. 100 (2): 429–36. doi:10.1111/j.1471-4159.2006.04248.x. PMID 17241159.
- ^ McVeigh JJ, Lopaschuk GD (Oct 1990). "Dichloroacetate stimulation of glucose oxidation improves recovery of ischemic rat hearts". Am J Physiol. 259 (4 Pt 2): H1079-85. doi:10.1152/ajpheart.1990.259.4.H1079. PMID 2221115.
- ^ Jaimes, R 3rd (Jul 2015). "Functional response of the isolated, perfused normoxic heart to pyruvate dehydrogenase activation by dichloroacetate and pyruvate". Pflügers Arch. 468 (1): 131–42. doi:10.1007/s00424-015-1717-1. PMC 4701640. PMID 26142699.
{{cite journal}}: CS1 maint: numeric names: authors list (link)