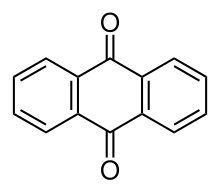
Anthraquinone
| |
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Anthracene-9,10-dione[2] | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| 390030 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.408 |
| 102870 | |
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 3143 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C14H8O2 | |
| Molar mass | 208.216 g·mol−1 |
| Appearance | Yellow solid |
| Density | 1.438 g/cm3[1] |
| Melting point | 284.8 °C (544.6 °F; 558.0 K)[1] |
| Boiling point | 377 °C (711 °F; 650 K)[1] |
| Insoluble | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
possible carcinogen |
| GHS labelling: | |

| |
| Danger | |
| H350 | |
| P201, P202, P281, P308+P313, P405, P501 | |
| Flash point | 185 °C (365 °F; 458 K) |
| Related compounds | |
Related compounds
|
quinone, anthracene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Anthraquinone, also called anthracenedione or dioxoanthracene, is an aromatic organic compound with formula C
14H
8O
2. Several isomers exist but these terms usually refer to 9,10-anthraquinone (IUPAC: 9,10-dioxoanthracene) wherein the keto groups are located on the central ring. It is used as a digester additive to wood pulp for papermaking. Many anthraquinone derivatives are generated by organisms or synthesised industrially for use as dyes, pharmaceuticals, and catalysts. Anthraquinone is a yellow, highly crystalline solid, poorly soluble in water but soluble in hot organic solvents. It is almost completely insoluble in ethanol near room temperature but 2.25 g will dissolve in 100 g of boiling ethanol. It is found in nature as the rare mineral hoelite.
Synthesis
[edit]There are several current industrial methods to produce 9,10-anthraquinone:
- The oxidation of anthracene. Chromium(VI) is the typical oxidant.
- The Friedel-Crafts reaction of benzene and phthalic anhydride in presence of AlCl3. o-Benzoylbenzoic acid is an intermediate. This reaction is useful for producing substituted anthraquinones.
- The Diels-Alder reaction of naphthoquinone and butadiene followed by oxidative dehydrogenation.
- The acid-catalyzed dimerization of styrene to give a 1,3-diphenylbutene, which then can be transformed to the anthraquinone.[3] This process was pioneered by BASF.
It also arises via the Rickert–Alder reaction, a retro-Diels–Alder reaction.
Reactions
[edit]Hydrogenation gives dihydroanthraquinone (anthrahydroquinone). Reduction with copper gives anthrone.[4] Sulfonation with sulfuric acid gives anthroquinone-1-sulfonic acid,[5] which reacts with sodium chlorate to give 1-chloroanthaquinone.[6]
Applications
[edit]Digester additive in papermaking
[edit]9,10-Anthraquinone is used as a digester additive in production of paper pulp by alkaline processes, like the kraft, the alkaline sulfite or the Soda-AQ processes. The anthraquinone is a redox catalyst. The reaction mechanism may involve single electron transfer (SET).[7] The anthraquinone oxidizes the reducing end of polysaccharides in the pulp, i.e., cellulose and hemicellulose, and thereby protecting it from alkaline degradation (peeling). The anthraquinone is reduced to 9,10-dihydroxyanthracene which then can react with lignin. The lignin is degraded and becomes more watersoluble and thereby more easy to wash away from the pulp, while the anthraquinone is regenerated. This process gives an increase in yield of pulp, typically 1–3% and a reduction in kappa number.[8]
Niche uses
[edit]9,10-anthraquinone is used as a bird repellant on seeds, and as a gas generator in satellite balloons.[9] It has also been mixed with lanolin and used as a wool spray to protect sheep flocks against kea attacks in New Zealand.[10]
Other isomers
[edit]Several other isomers of anthraquinone exist, including the 1,2-, 1,4-, and 2,6-anthraquinones. They are of minor importance compared to 9,10-anthraquinone.
Safety
[edit]Anthraquinone has no recorded LD50, probably because it is so insoluble in water.
In terms of metabolism of substituted anthraquinones, the enzyme encoded by the gene UGT1A8 has glucuronidase activity with many substrates including anthraquinones.[11]
See also
[edit]References
[edit]- ^ a b c d Haynes, William M., ed. (2016). CRC Handbook of Chemistry and Physics (97th ed.). CRC Press. p. 3.28. ISBN 9781498754293.
- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 724. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ Vogel, A. "Anthraquinone". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_347. ISBN 978-3527306732.
- ^ Macleod, L. C.; Allen, C. F. H. (1934). "Benzanthrone". Organic Syntheses. 14: 4. doi:10.15227/orgsyn.014.0004.
- ^ Scott, W. J.; Allen, C. F. H. (1938). "Potassium Anthraquinone-α-Sulfonate". Organic Syntheses. 18: 72. doi:10.15227/orgsyn.018.0072.
- ^ Scott, W. J.; Allen, C. F. H. (1938). "α-Chloroanthraquinone". Organic Syntheses. 18: 15. doi:10.15227/orgsyn.018.0015.
- ^ Samp, J. C. (2008). A comprehensive mechanism for anthraquinone mass transfer in alkaline pulping (Thesis). Georgia Institute of Technology. p. 30. hdl:1853/24767.
- ^ Sturgeoff, L. G.; Pitl, Y. (1997) [1993]. "Low Kappa Pulping without Capital Investment". In Goyal, G. C. (ed.). Anthraquinone Pulping. TAPPI Press. pp. 3–9. ISBN 0-89852-340-0.
- ^ "www.americanheritage.com". Archived from the original on 2009-06-09. Retrieved 2009-09-22.
- ^ Dudding, Adam (29 July 2012). "How to solve a problem like a kea". Sunday Star Times. New Zealand. Retrieved 11 November 2014.
- ^ Ritter, J. K.; Chen, F.; Sheen, Y. Y.; Tran, H. M.; Kimura, S.; Yeatman, M. T.; Owens, I. S. (1992). "A Novel Complex Locus UGT1 Encodes Human Bilirubin, Phenol, and other UDP-Glucuronosyltransferase Isozymes with Identical Carboxyl Termini" (PDF). Journal of Biological Chemistry. 267 (5): 3257–3261. doi:10.1016/S0021-9258(19)50724-4. PMID 1339448.
